Our new Associate Editor, Isabel Smallegange, is an Associate Professor of Population Biology at the University of Amsterdam. Follow her @I_Smallegange or check out her blogs at isabelsmallegange.com, where she also discusses issues regarding work-life and gender balance in academia.

In her research, Isabel takes the existing body of ecological and evolutionary theory and applies it to ecological datasets to understand how ecological and evolutionary processes interact.
During her PhD, Isabel specialised in behavioural ecology, but she moved into the field of population biology and demography (to model and empirically test the eco-evolutionary dynamics of populations) during her Post Docs at the Max Planck Institute for Ornithology, Imperial College and Oxford (see her papers on The stochastic demography of two coexisting male morphs and Towards a general, population-level understanding of eco-evolutionary change [1-2].
Two papers published in Functional Ecology were instrumental in this move. Firstly, the 1989 essay review by Stephen C. Stearns on trade-offs in life-history evolution [3] still provides valuable and useful insights into understanding the evolution of life history trade-offs, and also formed the basis for her to write a chapter on life history trade-offs in the Encyclopedia of Evolutionary Biology [4]. Secondly, the paper by Gregor Fussmann and co-workers [5] was a crucial review paper for getting to grips with eco-evolutionary dynamics.
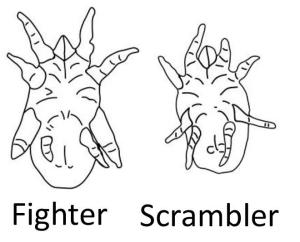
Now at the University of Amsterdam, Isabel has two main lines of research. In the first one, she examines the coexistence and maintenance of discrete alternative phenotypes within single populations, both in theory [6] and in experiments [7,8], thereby focusing on the alternative mating phenotypes of the bulb mite Rhizoglyphus robini (Figure 1). Such alternative phenotype systems present ideal test beds to investigate eco-evolutionary feedback loops (Figure 2), of which we yet understand very little.

Her second research line focuses on developing theory for accurately predicting eco-evolutionary population change [9-10]. The first steps towards this goal were to integrate dynamic energy budget theory into integral projection models [9], but many challenges still remain, like including quantitative genetics and interference competition [10], and empirically teasing apart both pathways within an eco-evolutionary feedback loop. A 2012 paper by Claus-Peter Stelzer, published in Functional Ecology [11], is one of the few studies to have done so. Unravelling eco-evolutionary feedback loops within single populations should lead to a solid understanding of how an environmental perturbation, such as a change in precipitation, temperature, predation pressure, etc., affects the two pathways of an eco-evolutionary loop: does a perturbation mainly affect ecological variables, with knock-on evolutionary consequences through the ecology-to-evolution pathway, or would it mainly result in an evolutionary shift in trait expression? Or perhaps both are affected simultaneously? These are essential insights to understand the dynamics of the natural world around us, and how they respond to the ever greater changes in the environment.
If you are a postdoctoral researcher with a quantitatively-driven mind, with interests in eco-evolutionary dynamics, life history, or demography and have a project in mind that you would like to develop in Isabel’s group, please contact her with a brief project proposal, CV and list of funding themes that you are considering for this project (e.g. Marie Curie, NWO-VENI).
References
-
- Smallegange IM, Coulson T. 2011. The stochastic demography of two coexisting male morphs. Ecology 92:755-764.
- Smallegange IM, Coulson T. 2013. Towards a general, population-level understanding of eco-evolutionary change. Trends in Ecology & Evolution 28:143-148.
- Stearns SC. 1989. Trade-offs in life-history evolution. Functional Ecology 3: 259-268.
- Smallegange IM. 2016. Life History Trade-offs. In: Kliman, R.M. (ed.), Encyclopedia of Evolutionary Biology. Vol. 2, pp. 390–393. Oxford: Academic Press.
- Fussmann GF, Loreau M, Abrams P.A. 2007. Eco-evolutionary dynamics of communities and ecosystems. Functional Ecology 21: 465-477.
- Smallegange IM, Johansson J. 2014. Life history differences favour evolution of male dimorphism in competitive games. American Naturalist 183: 188-198.
- Smallegange IM, Deere JA. 2014. Eco-evolutionary interactions as a consequence of selection on a secondary sexual trait. Advances in Ecological Research 50: 145-169.
- Smallegange IM, Fernandes RE, Croll JC. 2018. Population consequences of individual heterogeneity in life histories: overcompensation in response to harvesting of alternative reproductive tactics. Oikos 127: 738-749.
- Smallegange IM, Caswell H, Toorians MEM, de Roos AM. 2017. Mechanistic description of population dynamics using dynamic energy budget theory incorporated into integral projection models. Methods in Ecology and Evolution 8: 146-154.
- Smallegange IM, Ens HM, 2018. Trait-based predictions and responses from laboratory mite populations to harvesting in stochastic environments. Journal of Animal Ecology 87: 893-905.
- Stelzer C-P. 2012. Population regulation in sexual and asexual rotifers: an eco-evolutionary feedback to population size? Functional Ecology 26: 180-188.
